
LIFEPAK
®
15
monitor/defibrillator
Instructor guide

Contents
How to use this guide ...................................................................................................1
Training tips .....................................................................................................................1
Early debrillation ........................................................................................................ 2
Biphasic energy ...............................................................................................................2
Device power ....................................................................................................................3
Batteries ............................................................................................................................ 3
AC power ........................................................................................................................... 4
DC power ..........................................................................................................................5
Loading paper ..................................................................................................................6
Device maintenance ...................................................................................................... 7
AED training course ....................................................................................................11
Learning objectives ........................................................................................................11
Equipment and materials ...............................................................................................11
LIFEPAK 15 monitor/debrillator AED training class ..................................................11
Controls and features .................................................................................................12
AED operation................................................................................................................13
Manual training course .............................................................................................15
Learning objectives ........................................................................................................15
Equipment and materials ...............................................................................................15
LIFEPAK 15 monitor/debrillator Manual User Training Class ................................... 16
Controls and features .................................................................................................17
Connectors for IP monitoring conguration .................................................................23
Connectors for temperature monitoring conguration.................................................24
Back view ........................................................................................................................25
Home screen ...................................................................................................................26
Manual debrillation .................................................................................................27
Synchronized cardioversion procedure ..............................................................30
Noninvasive pacing .....................................................................................................32
ECG monitoring ............................................................................................................. 34
Acquiring a 12-lead ECG ............................................................................................37
SpO
2
, SpCO, and SpMet monitoring .......................................................................38
Pulse Oximetry ..............................................................................................................38
SpCO and SpMet ............................................................................................................39
Keys to successful SpCO and SpMet monitoring .............................................40
Monitoring noninvasive blood pressure ............................................................. 41
Monitoring EtCO
2
.........................................................................................................42
Monitoring Invasive Pressure .................................................................................43
Monitoring continuous temperature ...................................................................44
Vital signs and ST segment trends ........................................................................44
Data management ........................................................................................................47
Other functions .............................................................................................................49
Instructor guide | 1
How to use this guide
The LIFEPAK 15 monitor/debrillator is easy to use and enables clinicians at every level
to focus on the most important task at hand—saving a patient’s life. The LIFEPAK 15
device is highly intuitive to use, and adapts to various patient environments. With this
comprehensive guide, you’ll be able to train your staff to effectively use the device.
This instructor guide is an introduction to the basic operation of the LIFEPAK 15 device.
It does not suggest protocols or policies regarding the use of the debrillator. Refer
to the Operating Instructions for complete directions for use, indications,
contraindications, warnings, precautions and potential adverse events.
This outline has been designed for factory default conguration based on the 2010
American Heart Association Guidelines. It is important to be familiar with the
conguration of your particular debrillator. Moving, removing, highlighting and
adding content to this outline to meet individual user needs is encouraged. Hands-on
practice and application with scenarios promote learning retention.
Training tips
This guide is divided into ve sections:
Device power, paper change and maintenance
AED operation
Manual operation
Optional features
Data management and other functions
The AED mode is easy for healthcare professionals of all skill levels to quickly understand
and use. This training course reviews the basic operation of the LIFEPAK 15 device in AED
mode. The manual mode operation is for ALS providers and reviews the manual operation
and functions of the LIFEPAK 15 device.
The LIFEPAK 15 device retains data for two or more patients when you switch the power
off or remove the batteries. The device automatically stores a CODE SUMMARY™ report
as part of the patient report for each patient. This section describes how to access this
information and other functions associated with the LIFEPAK 15 device.
All sections are optimally taught in a hands-on format. Instructors should rst demonstrate
how to use the debrillator and then have students practice. Ideally, students will receive
enough practice and coaching from the instructor to ensure they can use the device with
condence in an actual emergency.
2 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Early defibrillation
A recent New England Journal of Medicine study of 6,789 in-hospital cardiac arrest events
due to ventricular brillation or pulseless ventricular tachycardia reported 70% received
debrillation in 2 or less minutes from recognition of Cardiac Arrest (CA). The survival rate
(signicantly) declined for the 30% of the patients who received debrillation more than
two minutes after cardiac arrest.
1
The American Heart Association (AHA) recommends debrillation within 2 minutes or less
of collapse in the hospital setting. The AHA suggests hospitals deploy AEDs throughout the
hospital to achieve this target and train staff to use the AEDs.
AEDs can help your hospital improve its ability to deliver the rst shock within the
recommended 2 minutes or less guidelines. The LIFEPAK 15 monitor/debrillator can be
used in AED mode to debrillate without having to learn ECG interpretation. The AED is
simple to use because it is designed to automatically analyze the patient’s heart rhythm
and advise you which steps to take. In AED mode the device automatically selects the
appropriate energy dosing.
Biphasic energy
Healthcare providers may have questions about recommended energy dosing for biphasic
waveforms because different debrillator manufacturers recommend different energy
dosing protocols. It is important to clarify the correct recommended energy dose for
biphasic waveforms in order to avoid possible confusion that may result in a delay
of therapy. Biphasic technology provides the option to escalate to 360J for best results.
Studies have shown that rebrillation is common among ventricular brillation
(VF) CA patients and that debrillation of recurring episodes of VF is increasingly
difcult.
2,3,4
LIFEPAK devices give you the option to escalate your energy dosing up
to 360J for difcult-to-debrillate patients.
Fortunately, all AEDs are programmed to follow a predened energy dosing protocol.
In other words, the healthcare provider doesn’t have to worry about energy dosing.
The AED performs this automatically.
A biphasic waveform sends current one way at the start of the shock and then reverses
it so the current ows in the opposite direction. Stryker recommends a dosing protocol
of 200-300-360 Joules and this is the factory default setting in all LIFEPAK debrillators.
Stryker believes this energy protocol can help minimize unnecessary CPR interruptions
that result from ineffective debrillation shocks.

Device power,
Paper change, and
Maintenance

Instructor guide | 3
Device power
The LIFEPAK 15 monitor/debrillator operates either on battery power using two Lithium-
ion batteries or with auxiliary power using the AC Power Adapter or DC Power Adapter.
Batteries
The LIFEPAK 15 device uses lithium-ion batteries with a typical operating time for two
new fully charged batteries of 360 minutes for monitoring, 340 minutes pacing or for
420 360 Joule shocks. The batteries can be recharged in the device if connected to an
approved AC or DC power adapter or by removing the batteries and placing them in an
approved battery charger. Batteries may be charged in the Station and Mobile Li-ion
Battery Charger, the REDI-CHARGE
®
battery charger, or in the monitor/debrillator
if it is connected to auxiliary power (AC or DC power adapter).
Each battery has a fuel gauge that indicates the approximate charge level in the battery.
Press the gray button above the battery symbol to check the battery’s charge level prior
to installing it in the debrillator. The four battery indicators shown here represent
approximate charge—greater than 70%, greater than 50%, greater than 25%, and 25%
or less, respectively.
When the LIFEPAK 15 device is turned on the Home Screen displays battery indicators
that show the following information about the batteries installed in the debrillator:
• Presence or absence of battery in battery well
• Battery in use
• Battery charge state
When two batteries are installed, the debrillator uses the battery with the lowest level
of charge rst. The battery in use is indicated by a white battery number in a black box.
When a battery reaches the replace battery state, the debrillator automatically switches
to the other battery.

4 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Indicator Meaning Description
Active battery
The debrillator is using the battery in well 1 for
power. Battery status indicators display up to four
green bars. Each green bar represents approximately
25% remaining charge. For example, three green bars
indicate about 75% remaining charge.
Low battery
Battery in well 1 is in use and is low. One yellow
bar indicates 5% to 10% remaining charge.
Very low
battery
Battery in well 1 is in use and is very low. One red
ashing bar indicates 0 to 5% remaining charge.
The debrillator automatically switches to the other
battery only if adequate charge is available. If both
batteries show red bars, the REPLACE BATTERY voice
prompt occurs.
Unrecognized
battery
Battery in well 2 is not in use. Battery communication
failed or a non-Stryker battery is installed. The battery
may power the debrillator but the level of charge
is unknown and low battery messages and prompts
will not occur.
No battery
installed or
fault detected
No battery is installed in battery well 1, or a fault was
detected in the battery in well 1 and the device will not
use the battery.
AC power
To use AC power;
1. Connect the AC power cord to the power adapter and a grounded AC outlet
2. Conrm LED strip on power adapter is illuminated
3. Connect power adapter output cable to power adapter
4. Connect green end of output cable to auxiliary power connector
on back of debrillator
5. Conrm
and LEDs are illuminated
Note: At least one battery should be installed at all times. Keep monitor/debrillator
connected to auxiliary power whenever possible to maintain battery charge level.
Instructor guide | 5
LED is illuminated whenever connected to auxiliary power, whether
debrillator is on or off
LED is illuminated if batteries are fully charged. LED ashes if either
battery is being charged
LOW BATTERY and REPLACE BATTERY warning messages do not occur when
connected to auxiliary power
To quickly determine whether power source is battery or auxiliary power, either:
• Check LED. Illuminated LED indicates auxiliary power in use; or
• Check whether battery well number is highlighted. Highlighted battery well
number indicates battery in use.
Battery Charging Indicator Behaviors
Steady green Installed batteries are fully charged.
Flashing green One or both installed batteries are being charged.
Off No batteries are installed or a battery is unable to be charged.
DC power
To use the DC Power Adapter:
1. Connect the DC power cable to the power adapter and a 12 Vdc power source.
2. Verify that the green LED strip illuminates.
3. Connect the power adapter output cable to the power adapter.
4. Connect the green end of the power adapter output cable to the auxiliary power
connector on the back of the monitor/debrillator.
5. Verify that the AUXILIARY POWER LED on the debrillator is illuminated.
6. If at least one battery is installed in the device, verify that the BATTERY
CHARGING indicator is illuminated or ashing. Indicator behaviors are
shown in the Table above.
Battery LED
6 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Loading paper
Check the amount of paper in the printer as part of the daily check according to the
Operator’s Checklist provided in the back of the Operating Instructions.
The printer is equipped with an out-of-paper sensor to protect the printer printhead. The
sensor automatically turns off the printer if paper runs out or the printer door is open.
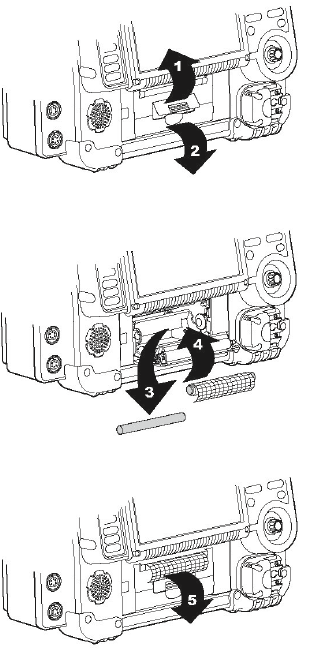
To load paper:
1. Lift the printer door latch to release the door (see Figure 10-1).
2. Pull out the printer door.
3. Remove the empty paper spool, if present.
4. Insert a new paper roll with the graph side facing up. Make sure the end of
the paper extends outward so it is exposed when the printer door is closed.
5. Close the printer door and press down on the latch until the door clicks shut.

Instructor guide | 7
Device maintenance
Most hospitals and EMS services have specic protocols for maintaining their
debrillators. Stryker provides a variety of tools designed to help manage inspection and
maintenance procedures. Training for staff would depend on a hospital’s or EMS service’s
particular approach.
BLS-trained responders should be familiar with several of the device maintenance
procedures. How much detail you choose to go into will depend on your hospital’s protocols.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Check debrillator
for Daily Auto
Test results, if
congured on.
User actions
• Ensure that the batteries are fully charged.
• Ensure that the debrillator is plugged into the AC
power adapter and that the adapter is plugged into
electricity, if applicable.
• Ensure that the QUIK-COMBO
®
therapy cable is
connected to the debrillator.
or
• Ensure the standard paddles are clean, dry and
properly seated in the paddle wells and connected
to the debrillator.
If the debrillator detects a problem during the
self-test the printed report will indicate SELF TEST
FAILED. The SERVICE LED will illuminate the next
time it is turned on.
Debrillator should be checked daily following the
auto test to conrm the test passed. If test failed or
incomplete perform the manual user test.
Task
Perform QUIK-
COMBO therapy
cable check in
Manual mode:*
User actions
1. Disconnect and examine cable for cracking,
damage, broken, or bent parts or pins.
2. Connect therapy cable to debrillator and the
Test Load.
3. Select 200 JOULES and press CHARGE.
4. Press
(SHOCK) button.
5. Conrm ENERGY DELIVERED message appears.
6. Remove the Test Load from cable.**
(PADDLES LEADS OFF appears if Paddles
lead displayed.)
* The debrillator delivers up to 360 Joules of
electrical energy. Unless discharged properly, this
electrical energy may cause serious personal injury
or death. Do not attempt to perform this test unless
you are qualied by training and experience.
** Failure to remove the Test Load may result in
delay of therapy during patient use.
Instructor activity
Conrm test load
is connected to
QUIK-COMBO cable.

8 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Task
Perform standard
(hard) paddles check
in Manual mode.*
User actions
1. Disconnect and examine cable for cracking,
damage, broken or bent parts or pins.
2. Connect paddles to debrillator.
3. Examine for paddle surface pitting and presence
of dried or wet gel.
4. Press LEAD. Select Paddles.
5. On paddles, turn ENERGY SELECT dial
to 10 JOULES.
6. With paddles in paddle wells, press CHARGE
button on paddle.
7. Press only one (SHOCK) button and release.
Conrm that energy was not discharged.
8. Press the other (SHOCK) and release. Conrm
that energy was not discharged.
9. Press both (SHOCK) and conrm ABNORMAL
ENERGY DELIVERED message appears.
10. Remove paddles from wells, and conrm artifact
on screen.
11. Place paddle surfaces together, and conrm at
line on screen.
12. Return paddles securely to paddle wells.
Task
Inspect the physical
condition of
the debrillator.
User action
Inspect debrillator for damage and
foreign substances.
Task
Inspect
power source.
User actions
• Conrm that batteries are fully charged.
• Conrm that Auxiliary Power Indicator
is on if applicable.
Task
Check therapy and
ECG electrodes.
User actions
Check QUIK-COMBO and EGG electrodes for
“use by” date and that a spare set is available.
Task
Examine
accessory cables.
User actions
Inspect all cables including power cord for cracks,
broken or bent parts and pins, and, if applicable,
paddle surfaces for pitting.
Task
Disconnect
debrillator from
AC power.
Press ON.
Look for SELF-
TEST message and
illumination of LEDs.
User actions
Debrillator turns on.
• If debrillator doesn’t turn on, contact qualied
service personnel.
• There should not be low battery or replace battery
messages visible. If visible, it means one or both
batteries need to be charged.
Instructor activity
This only applies to
devices with the AC
Power Adapter.

Instructor guide | 9
Task
Perform Manual
User Test.
Press ON.
• Press OPTIONS.
• Select USER TEST.
User actions
• Conrm AC MAINS LED is lit, if applicable.
• User action, debrillator labels, text/voice prompts
and information
• The manual user test should be performed if your
protocol requires more frequent testing than the
recommended daily auto test, or the daily auto test
failed or did not complete.
Note: If the debrillator is in AED mode,
switch to manual mode by Pressing the
ENERGY SELECT button.
Device will print the test results when
the test completes.
If the user test fails, contact qualied
service personnel.
Task
Cleaning
User actions
Clean the LIFEPAK 15 monitor/debrillator, cables and
accessories with a damp sponge or cloth. Do not use
bleach. Use only the agents listed below:
• Quarternary ammonium compounds
• Isopropyl alcohol
• Peracetic (peroxide) acid solutions
10 | Instructor guide for the LIFEPAK 15 monitor/debrillator/

AED operation
Instructor guide | 11
AED training course
The AED mode is highly intuitive and easy for healthcare professionals of all skill levels
to quickly understand and use. This training course reviews the basic operation of the
LIFEPAK 15 device in AED mode.
Learning objectives
The overall objective of this inservice is to provide an overview of the basic steps of
operation of the identied controls, indicators and connectors of the LIFEPAK 15 device.
Upon completion of this course, participants will be able to:
• Verbalize the importance of early debrillation.
• Locate and identify the debrillator’s front panel controls, indicators and connectors.
• Demonstrate QUIK-COMBO electrode application.
• Demonstrate steps for automated external debrillation (AED).
• Demonstrate the paper change.
• Demonstrate routine daily testing.
Equipment and materials
The following is a list of accessories and support material recommended for training
on the LIFEPAK 15 device. It is essential that all equipment be inspected and tested to
ensure proper function prior to training according to the Basic Orientation section of the
Operating Instructions.
Equipment
• LIFEPAK 15 monitor/debrillator
Accessories
• QUIK-COMBO therapy cable
• Test Load
• EDGE System™ electrodes with QUIK-COMBO connector (or clip-on training electrodes)
• QUIK-COMBO 3-lead or 12-lead patient simulator
• AC power adapter and cord if applicable
• ECG printer paper
Support materials
• Quick Reference Cards
• Self Assessment Form for BLS Users
• Operating Instructions
• Performance Evaluations
LIFEPAK 15 monitor/debrillator AED training class
The following lists the content that should be covered during a typical AED
operation training class.
• Early debrillation
• Biphasic technology
• Controls and features
• AED operation
• Device maintenance and power
12 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
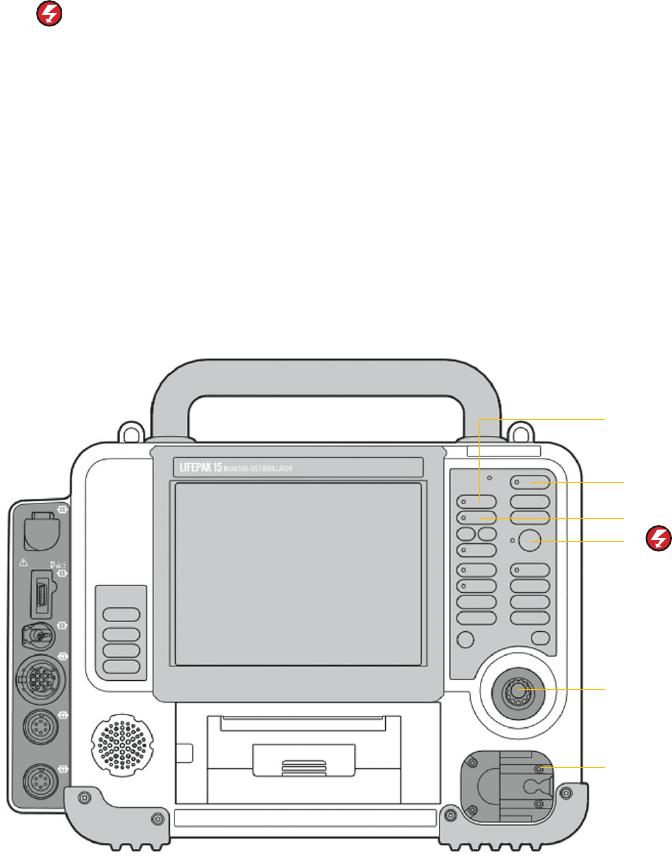
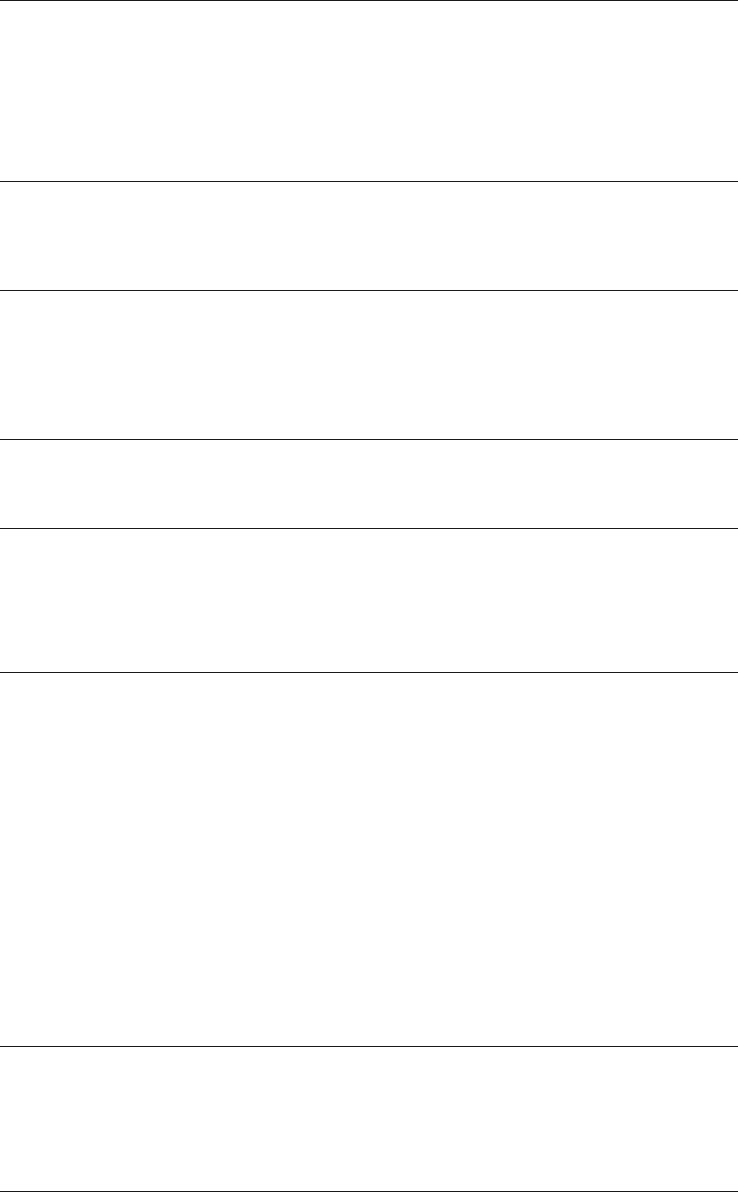
Controls and features
In this section, the goal is to point out the different buttons and physical features pertinent
to an AED user. Each of these features will be reviewed in greater detail later in this guide.
Refer to the Operating Instructions for additional information.
AED buttons
Three buttons used for AED operation.
1. ON
2. ANALYZE
3.
CPR
Used to turn the metronome off and on.
Speed dial
Scrolls through and selects menu items.
Therapy cable connector
Connects therapy cable to the device.
Therapy cable (not shown)
The therapy cable is a debrillation cable that attaches to therapy electrodes.
The LIFEPAK 15 device should be stored with the therapy cable plugged into the
lower right-hand side of the device.
CPR
AED buttons
1. ON
2. ANALYZE
3.
(SHOCK)
Speed dial
Therapy cable
connector
Instructor guide | 13
AED operation
The AED mode on the LIFEPAK 15 device is easy to use because it automatically analyzes
the patient’s heart rhythm and advises you which steps to take. In AED mode the device
automatically selects the appropriate energy dosing.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions.
Task
Verify the patient is
in cardiopulmonary
arrest: unconscious/
unresponsive, not
breathing normally,
and showing no signs
of circulation.
Press ON.
User action, debrillator labels, text/voice
prompts and information
Note the CONNECT ELECTRODES message and
voice prompt occurs until the patient is connected
to the AED.
Task
Prepare the
patient for therapy
electrode placement.
Connect therapy
electrodes to the
therapy cable,
and conrm cable
connection to the
debrillator.
User action, debrillator labels, text/voice
prompts and informatio
1. Bare patient’s chest.
2. Remove excessive chest hair.
3. Clean and dry skin.
4. Abrade the skin briey using a dry towel or
gauze for better electrode adhesion to the skin.
• Do not use alcohol, tincture of benzoin or
antiperspirant to prepare the skin.
Instructor activity
Connect therapy
cable to the rhythm
simulator and set
rhythm to VF.
Task
Apply therapy
electrodes to
patient’s chest in
anterior-lateral
position.
Instructor activity
Conrm correct
placement.
Task
Press ANALYZE
button to initiate
analysis.
Stop CPR.
User actions
You will see and hear the PUSH ANALYZE message.
• Stop all motion including CPR.
• Do not move the LIFEPAK 15 device
while analyzing.
• Clear everyone away from patient.
Task
Follow screen
messages and
voice prompts.
Conrm everyone
is clear.
Press
(SHOCK)
button to
discharge AED.
User actions
You will see and hear ANALYZING NOW STAND
CLEAR, PUSH TO SHOCK followed by a “shock ready”
tone and ashing shock LED.
1. State “All Clear” and observe that all personnel
are clear of the patient and immediate area.
2. Press (SHOCK) button to discharge AED.
When the (SHOCK) button is pressed, you will see
Energy Delivered message indicating energy transfer
was completed.
Note: If the (SHOCK) button is not pressed
within 60 seconds, or if the SPEED DIAL is pressed
to cancel charging, the debrillator disarms and the
DISARMING message appears.
Note: When energy transfer is complete, the shock
counter increases by 1. This will continue to increase
incrementally with every energy transfer.
14 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Task
START CPR. analysis.
Stop CPR.
User action, debrillator labels, text/voice
prompts and information
START CPR screen message will appear.
• A CPR timer will countdown 2 minutes or CPR
time duration.
• A metronome automatically provides 30:2 audible
compression “tocks” and ventilation prompts. To
silence the metronome press CPR. To restart the
metronome, press CPR again.
Instructor activity
Place rhythm
simulator in
nonshockable
rhythm.
Task
The CPR metronome.
The CPR metronome provides audible “tocks” that
guide the user to deliver CPR with proper timing.
In AED Mode, the default C:V ratio is Adult – no
airway because most patients in cardiac arrest are
adults who have an initially unsecured airway.
Task
Stop CPR and
push ANALYZE.
User action, debrillator labels, text/voice
prompts and information
When the CPR countdown time ends, you will see and
hear PUSH ANALYZE.
This message stays on the screen and the voice prompt
will repeat every 20 seconds until the ANALYZE
button is pressed.
Task
Conrm everyone
is clear.
User action, debrillator labels, text/voice
prompts and information
If the AED detects a nonshockable rhythm, you will
see and hear NO SHOCK ADVISED.
Task
START CPR.
User action, debrillator labels, text/voice
prompts and information
Start CPR per voice prompt and screen message.
• A CPR timer will countdown 2 minutes and the
metronome will begin again.
• Continue to follow screen messages and voice
prompts until the advanced care team arrives.
Task
Troubleshooting
messages
User action, debrillator labels, text/voice
prompts and information
CONNECT ELECTRODES message and voice
prompt occur.
• If therapy electrodes are not connected
to the therapy cable.
or
• If therapy electrodes are not placed
on the patient’s chest.
CONNECT CABLE message occurs.
• If the therapy cable is not connected
to the debrillator.
MOTION DETECTED, STOP MOTION message occurs.
• If motion is detected during the ECG analysis, you
will see and hear MOTION DETECTED, STOP
MOTION, followed by a warning tone. Analysis is
delayed by no more than 10 seconds due to motion
detection. After 10 seconds, even if motion is still
present, the analysis proceeds to completion.
Task
Switching from
AED to
Manual Mode
Switching from
Manual Mode
to AED mode
User action, debrillator labels, text/voice
prompts and information
• Enter manual mode by pressing the ENERGY
SELECT button once to switch to manual mode.
• Pressing ANALYZE while the device is in manual
mode will return device to AED mode.
Instructor activity
Have students
switch device
between AED and
manual mode and
back to AED mode.

Manual operation
Manual training course
The manual mode training course is intended for the ALS-trained healthcare professionals
and reviews the manual operation and functions of the LIFEPAK 15 monitor/debrillator.
This training course reviews all of the advanced cardiovascular life support tools available
on the LIFEPAK 15 device.
Learning objectives
The overall objective of the inservice is to provide an overview of the basic steps of
operation of the identied controls, indicators and connectors of the LIFEPAK 15 device.
Upon completion of this course, participants will be able to:
• Verbalize the importance of early debrillation.
• Locate and identify the debrillator’s front panel controls, indicators and connectors.
• Demonstrate QUIK-COMBO electrode application.
• Demonstrate steps for manual debrillation.
• State the procedure for synchronized cardioversion.
• List the operational steps for noninvasive pacing.
• Demonstrate ECG, pulse oximetry monitoring and other optional features of the
LIFEPAK 15 device (if applicable).
• Describe the print process, and recalling the code summary.
• Explain the power sources.
• Demonstrate routine daily testing.
Equipment and materials
The following is a list of accessories and support material recommended for ACLS
training on the LIFEPAK 15 device. It is essential that all equipment be inspected
and tested to ensure proper function prior to training.
Equipment
• LIFEPAK 15 monitor/debrillator
Accessories
• QUIK-COMBO therapy cable
• Test Load
• EDGE System electrodes with QUIK-COMBO connector (or clip-on training electrodes)
• QUIK-COMBO 3-lead or 12-lead patient simulator
• 3-wire, 4-wire or 5-wire ECG cable
• 12-Lead cable and precordial leads (if applicable)
• LIFEPAK 15 monitor/debrillator standard adult detachable hard paddles (if applicable)
• Optional accessories (if applicable)
• SpO
2
or Rainbow
®
sensors
• EtCO
2
cannula and T-piece connector
• NIPB cuffs
• Temperature adapter and probes
• AC power adapter and cord (if applicable)
• 100 mm ECG printer paper
Instructor guide | 15
Support Materials
• Quick Reference Cards
• Self Assessment forms
• Operating Instructions
• Performance Evaluations
LIFEPAK 15 monitor/debrillator
Manual User Training Class
The following lists the content that should be covered during a typical
Manual User Training Class.
• Early debrillation
• Biphasic energy
• Controls and features
• Manual debrillation
• Synchronized cardioversion
• Noninvasive pacing
• ECG monitoring
• 12-Lead ECG acquisition and transmission (if applicable)
• Pulse oximetry or Rainbow technology (if applicable)
• EtCO
2
(if applicable)
• NIBP (if applicable)
• Temperature (if applicable)
• Invasive pressures (if applicable)
• Data management
• Device maintenance
16 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Instructor guide | 17
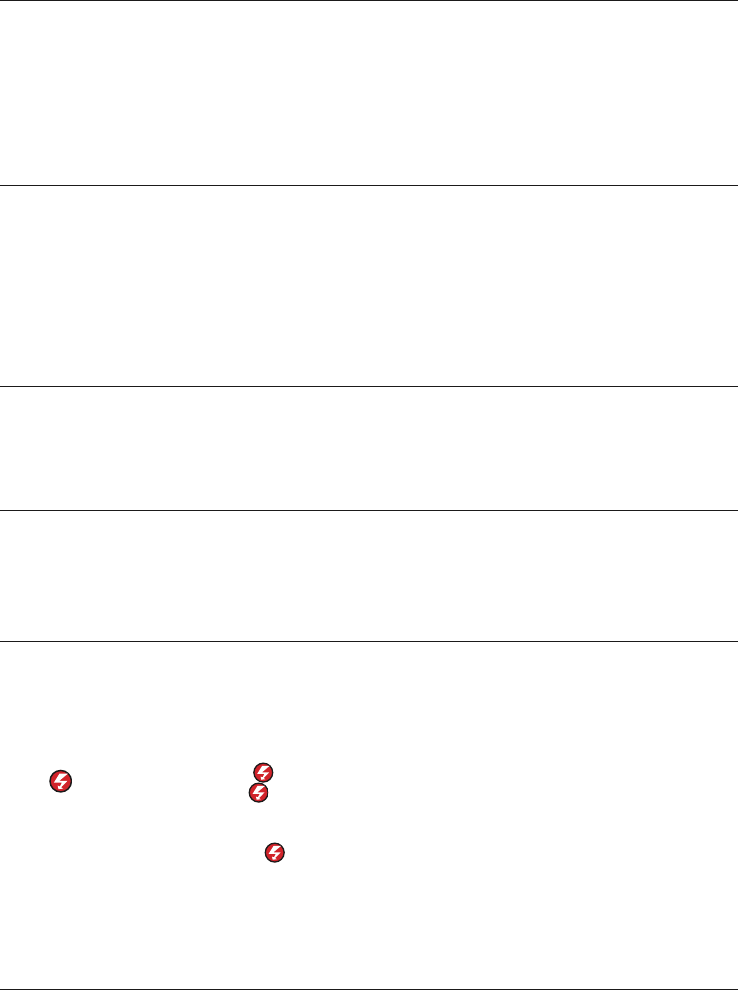
Controls and features
In this section, the goal is to point out the different buttons and physical features pertinent
to an ALS user. Each of these features will be reviewed in greater detail in latter sections
of this guide. For complete information, review the Operating Instructions for the device.
Area 1
Area 2
Area 3
Area 5
Area 4

ON
ENERGY
SELECT
CHARGE
CPR
ANALYZE
SIZE
SYNC
LEAD
SIZE
Changes ECG size
18 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
SYNC
Activates
synchronized mode
LEAD
Changes
ECG Lead
Buttons
• ON
• ENERGY SELECT
• CHARGE
•
(SHOCK)
• AC LED
• BATTERY LED
• SERVICE WRENCH
• CPR
• ANALYZE
• LEAD
• SIZE
• SYNC
Area 1
ON
Switches power
on or off
SHOCK
Discharges
debrillator
energy to the
patient
CHARGE
Charges the
debrillator in
manual mode
ENERGY
SELECT
Selects energy
levels in
manual mode
AC LED (graphic)
Illuminated AC LED indicates
device is plugged into electricity.
Battery LED (graphic)
Illuminated Battery LED indicated
battery charge status.
Service Wrench LED (graphic)
Illuminated Service LED indicates
service is required.
CPR
Controls CPR
metronome
ANALYZE
Activates
Shock
Advisory
System (SAS)

Instructor guide | 19
Area 2
Buttons
• PACER
• RATE
• CURRENT
• PAUSE
PACER
Activates the pacing function.
RATE
Selects pacing rate.
CURRENT
Adjusts pacing current.
PAUSE
Temporarily slows pacing rate.

NIBP
ALARMS
OPTIONS
EVENT
20 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Area 3
Buttons
• NIBP
• ALARMS
• OPTIONS
• EVENT
•
(Home screen)
• Speed Dial
•
HOME SCREEN
Returns
immediately to
Home Screen
NIBP
Initiates blood
pressure measurement
ALARMS
Activates and
silences alarms
OPTIONS
Accesses optional
functions
EVENT
Activates
user-dened
events
Speed Dial
Scrolls through
and selects screen
or menu items
Display mode
button switches
between color and
SunVue™ display

12-LEAD
TRANSMIT
CODE
SUMMARY
PRINT
Instructor guide | 21
Area 4
Buttons
• 12-LEAD
• TRANSMIT
• CODE SUMMARY
• PRINT
12-LEAD
Initiates acquisition of 12-lead ECG
TRANSMIT
Initiates transmission of patient data
CODE SUMMARY
Prints CODE SUMMARY critical event record
PRINT
Starts and stops printer

22 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
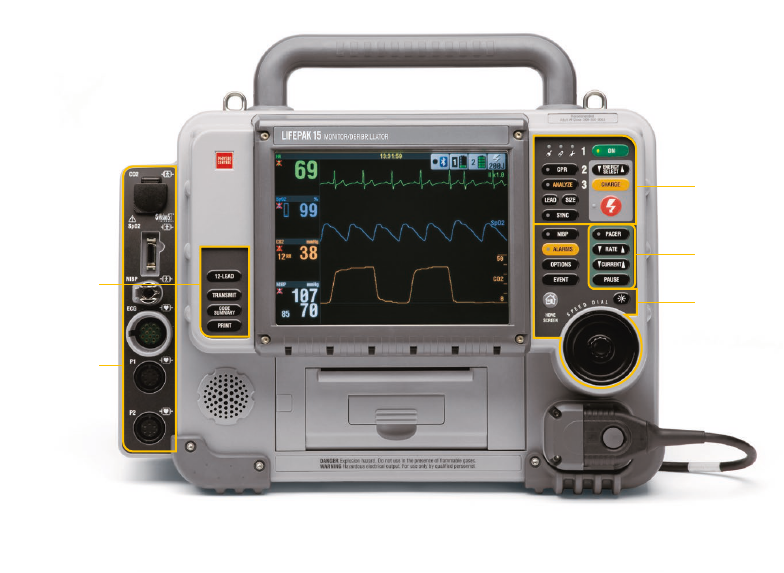
Area 5
• CO
2
• SpO
2
/SpCO/SpMet
• NIBP
• ECG
• P1
• P2
• TEMP
• Speaker
• Printer
• Therapy cable
receptacle
CO
2
Filter line
set port
Note: If your LIFEPAK 15 monitor/debrillator is congured for temperature monitoring,
P1 and P2 are replaced by a single port labeled TEMP.
Speaker
Projects device tones and voice prompts
Printer
Door for 100mm printer
Therapy Cable Receptacle
QUIK-COMBO therapy cable and standard
(hard) paddles cable receptacle
SpO
2
/SpCO/
SpMet
Sensor
cable port
NIBP
Pneumatic
tubing port
ECG
Green electrically isolated ECG cable port
P1
Invasive
pressure
cable port
P2
Invasive
pressure
cable port
Instructor guide | 23
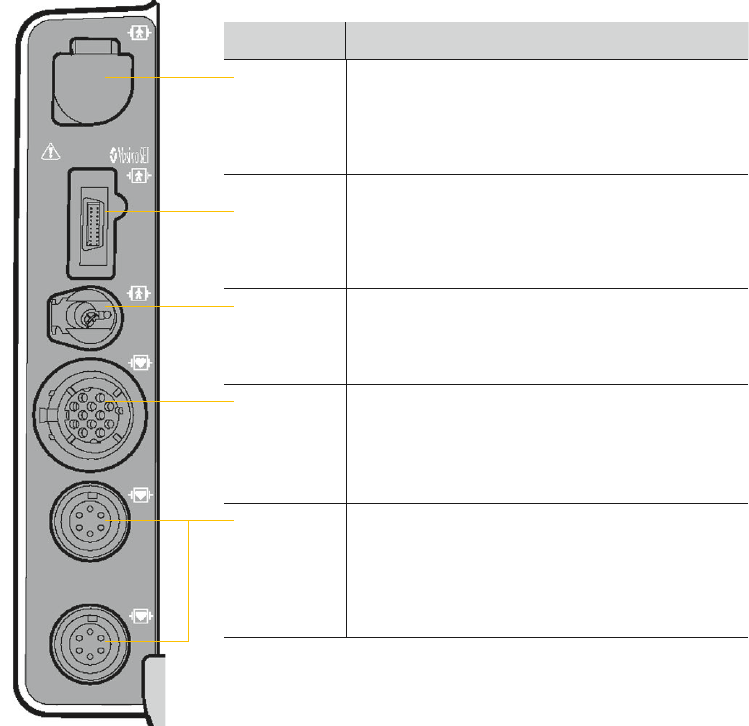
Connectors for IP monitoring conguration
Connector Action
CO
2
Connect: Open CO
2
port door, insert FilterLine
®
connector, and turn clockwise until connector is
rmly seated.
Disconnect: Rotate FilterLine connector
counterclockwise and pull connector out.
SpO
2
/
SpCO/
SpMet
Connect: Align cable connector with SpO
2
port and
push in until connector clicks into place.
Disconnect: Press the gray buttons on each side
of the cable connector simultaneously and pull
connector out.
NIBP Connect: Insert NIBP tubing connector into the
NIBP port.
Disconnect: Press the latch on the left side of the
port and pull tubing connector out.
ECG Connect: Align the green ECG connector with the
ECG port; position the white line on the cable facing
left. Insert the cable connector into the port until the
connector is rmly seated.
Disconnect: Pull the ECG connector straight out.
P1/P2 Connect: Align the IP (invasive pressure) cable
connector with the P1 or P2 port; position the gap on
the connector facing up. Insert the cable connector
into the port until the connector is rmly seated.
Disconnect: Grip the connector and pull
straight out.
24 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
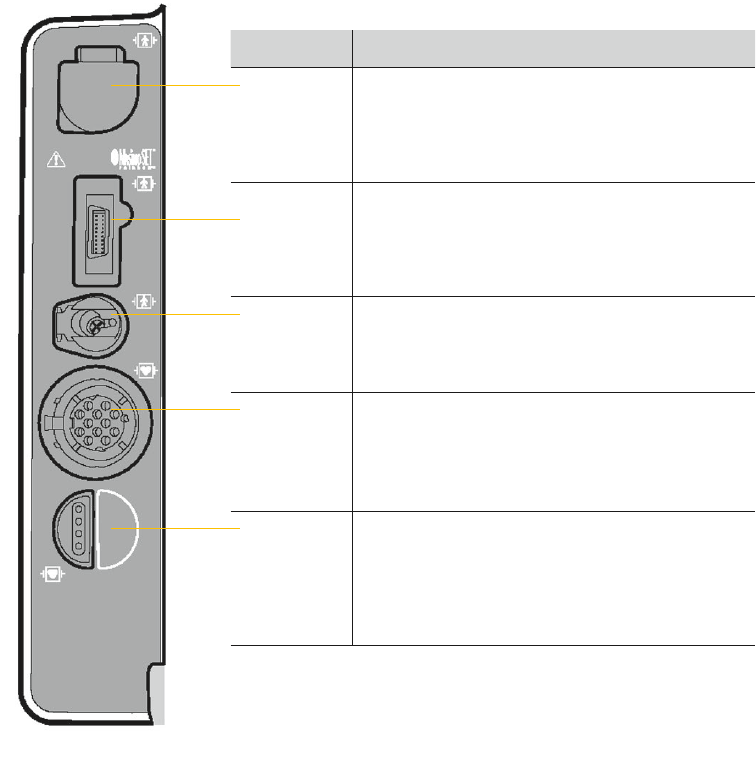
Connectors for temperature monitoring conguration
Connector Action
CO
2
Connect: Open CO
2
port door, insert FilterLine
connector, and turn clockwise until connector is
rmly seated.
Disconnect: Rotate FilterLine connector
counterclockwise and pull connector out.
SpO
2
/
SpCO/
SpMet
Connect: Align cable connector with SpO
2
port and
push in until connector clicks into place.
Disconnect: Press the gray buttons on each side
of the cable connector simultaneously and pull
connector out.
NIBP Connect: Insert NIBP tubing connector into the
NIBP port.
Disconnect: Press the latch on the left side of the
port and pull tubing connector out.
ECG Connect: Align the green ECG connector with the
ECG port; position the white line on the cable facing
left. Insert the cable connector into the port until the
connector is rmly seated.
Disconnect: Pull the ECG connector straight out.
TEMP Connect: Align the temperature adapter cable
connector with the TEMP port. Insert the cable
connector into the port until the connector is
rmly seated.
Disconnect: Grip the connector and pull
straight out.
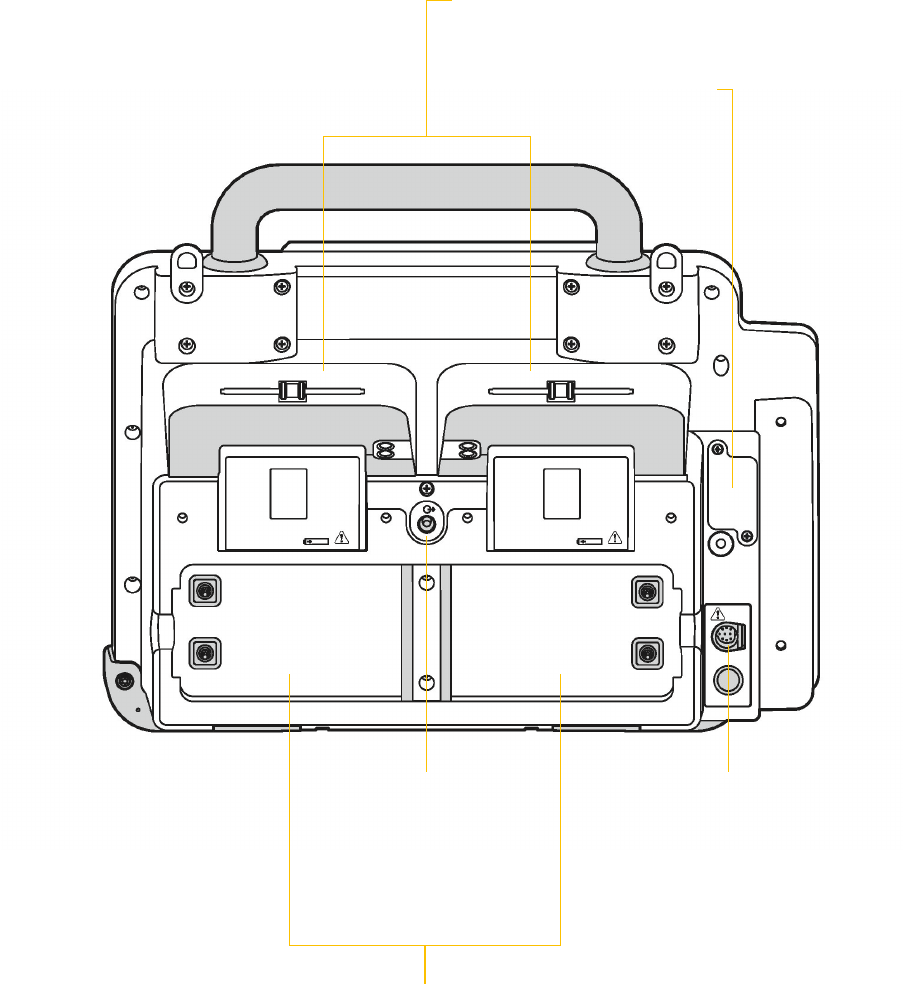
Instructor guide | 25
Back view
Battery wells
Each well holds on Lithium-ion battery
CO
2
exhaust port
Connects to
scavenger system
when monitoring
EtCO
2
during use
of anesthetics
Standard paddle wells
Paddle wells stow standard
(hard) paddles
USB port cover
Protect USB port
(Future use)
System connector
Connects the device to a gateway
or external computer for transfer
of patient data. Also provides
real-time ECG output.
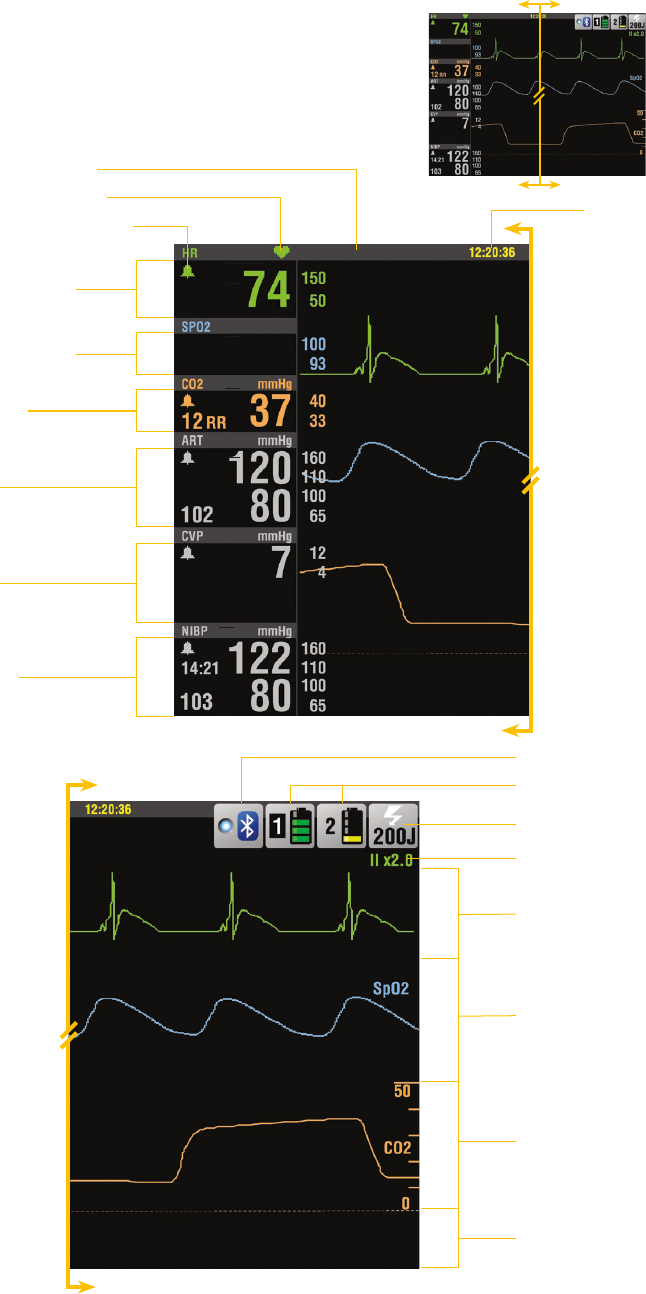
26 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Home screen
Time
Alarm limits
Heart symbol
Alarm indicator
Heart rate
SpO
2
/SpCO/
SpMet
EtCO
2
IP1
IP2
NIBP
Bluetooth icon
Battery indicator
Selected energy
ECG lead/size
Channel 1
Channel 2
Channel 3
Message area
Instructor guide | 27
Manual defibrillation
A direct current debrillator applies a brief, intense pulse of electricity to the heart muscle.
The LIFEPAK 15 monitor/debrillator delivers this energy through disposable electrodes,
standard paddles or internal paddles applied to the patient’s chest. Debrillation is only
one aspect of the medical care required to resuscitate a patient with a shockable ECG
rhythm. Depending on the situation, other supportive measures may include:
• Cardiopulmonary resuscitation (CPR)
• Administration of supplemental oxygen
• Drug therapy
Successful resuscitation is related to the length of time between the onset of a heart
rhythm that does not circulate blood (ventricular brillation, pulseless ventricular
tachycardia) and debrillation. The American Heart Association has identied the
following as critical links in the chain of survival from cardiac arrest:
1. Immediate recognition of cardiac arrest and activation of the
emergency response system
2. Early cardiopulmonary resuscitation (CPR) with an emphasis
on high-quality chest compressions
3. Rapid debrillation
4. Effective advanced life support
5. Integrated post-cardiac arrest care
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Verify the patient is
in cardiopulmonary
arrest: unconscious/
unresponsive, not
breathing normally,
and showing no signs
of circulation
Press ON.
Task
Connect therapy
electrodes to the
therapy cable,
and conrm cable
connection to the
debrillator.
Instructor activity
Connect therapy
cable to the
simulator and set
rhythm to VF.

28 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Task
Prepare the patient
for therapy electrode
placement.
Apply therapy
electrodes to
patient’s chest in
anterior-lateral
position.
Note: If needed,
refer to anterior-
lateral placement,
section 4 of
the Operating
Instructions.
User action, debrillator labels, text/voice
prompts and information
• Remove all clothing from the patient’s chest.
• Remove excessive chest hair.
• Clean and dry the skin.
• Do not use alcohol, tincture of benzoin, or
antiperspirant to prepare the skin.
• Ensure pads are in sealed package and the
use by date has not passed.
• Avoid placement over the nipple, bony
prominences, dressings, implantable
debrillators, or the diaphragm if possible.
• Apply therapy electrodes to patient’s chest
in anterior-lateral position.
• If using standard paddles, apply conductive
gel to the electrodes and place paddles on the
patient’s chest.
Note: Impedance is measured whenever the
debrillator is charged. To ensure therapeutic
patient impedance levels, you should always
charge the debrillator when the standard paddle
or QUIK-COMBO electrodes are in contact with
the patient’s chest.
Instructor activity
Demonstrate anterior
lateral position with
therapy electrodes
Task
Press ENERGY
SELECT.
User action, debrillator labels, text/voice
prompts and information
Select Joules per hospital-specic or training
protocols, if applicable.
Task
To change energy
selection.
User action, debrillator labels, text/voice
prompts and information
If energy selection is changed after charging has
started, the energy is removed. Press CHARGE
to restart charging.
Task
Press CHARGE.
Press SPEED DIAL
to disarm.
User action, debrillator labels, text/voice
prompts and information
While the debrillator is charging, a charging
bar appears and a ramping tone sounds,
indicating the charging energy level.
When debrillator is fully charged, an
overlay appears.
Task
Make certain all
personnel, including
the operator, stand
clear of the patient,
bed, and any
equipment connected
to the patient.
Press the
(SHOCK) button to
discharge energy to
the patient.
User action, debrillator labels, text/voice
prompts and information
State “All Clear” and observe that all personal
are clear of the patient and immediate area.
Conrm ECG rhythms and available energy.

Instructor guide | 29
Task
Press CPR button.
User action, debrillator labels, text/voice
prompts and information
1. Start CPR according to protocol.
2. To activate the metronome, press CPR.
3. Select the desired Age-Airway setting
using the SPEED DIAL.
4. Adult – No Airway 30:2 (default)
5. Adult – Airway 10:1
6. Youth – No Airway 15:2
7. Youth – Airway 10:1
8. Stop Metronome.
• To stop the metronome, select STOP
METRONOME from the CPR menu.
Discuss
The CPR metronome.
The CPR metronome provides audible “tocks” that
guide the user to deliver CPR with proper timing.
In Manual Mode, the user can choose the most
appropriate C:V ratio based on the patient’s age
and current airway status.

30 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Synchronized cardioversion procedure
The LIFEPAK 15 monitor/debrillator can be congured to remain in synchronous mode
or to return to asynchronous mode after discharge. It is important that you know how
your debrillator is congured.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Press ON.
Task
Attach patient ECG
cables and ECG
electrodes on
the patient.
Select Lead II
or the lead with
greatest QRS
complex amplitude
(positive or negative).
Observe the
ECG rhythm.
Press SYNC.
Observe the
ECG rhythm.
Prepare the patient’s
skin for therapy
electrode application.
Connect the
therapy electrodes
to the therapy
cable, conrm
cable connect
to the device.
Apply therapy
electrodes to the
patient in the
anterior-lateral
position. If using
standard paddles,
apply conductive gel
to the paddles and
place paddles on the
patient’s chest.
User action, debrillator labels, text/voice
prompts and information
• Bare patient’s chest.
• Ensure chest is clean and dry.
• Remove excessive chest hair.
• Prepare electrode site with brisk rub.
• Ensure electrodes are in sealed package and the
use by date has not passed.
• Avoid placement over the nipple, bony
prominences, dressings, implantable debrillators
or the diaphragm if possible.
Note: To monitor the ECG through therapy electrodes,
place the electrodes in the anterior-lateral position
and select paddles lead.
Conrm the Sync LED blinks with each detected
QRS complex.
Note: Press SYNC again to deactivate
synchronous mode.
Conrm that a triangle sense marker appears near the
middle of each QRS complex. If the sense markers do
not appear or are displayed in the wrong locations (for
example, on the T-wave), select another lead.
Instructor activity
Connect QUIK-
COMBO Therapy
Cable to the
simulator and set
rhythm to VT.
Task
Press
ENERGY SELECT.
User action, debrillator labels, text/voice
prompts and information
Select Joules per protocols, if applicable.
Task
Press CHARGE.

Instructor guide | 31
Task
Make certain all
personnel, including
operator, stand
clear of the patient,
bed, and any
equipment connect
to the patient.
Conrm rhythm and
available energy.
Task
Press and hold
(SHOCK)
button(s) until
you see ENERGY
DELIVERED
on screen.
User action, debrillator labels, text/voice
prompts and information
If (SHOCK) buttons are not pressed within
60 seconds, stored energy is internally removed.
Note: If the energy selection is changed after charging
has started, the energy is removed internally. Press
CHARGE to restart charging

32 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Noninvasive pacing
The noninvasive pacemaker can be used for either demand (synchronous) or nondemand
(asynchronous) pacing modes.
The demand mode is used for most patients. In the demand mode, the LIFEPAK 15
monitor/debrillator/pacemaker inhibits pacing when it senses the patient’s own
beats, if the ECG amplitude is too low to detect the patient’s beats or if an ECG lead
becomes detached so that the ECG rhythm is not present, the pacemaker generates
pacing pulses asynchronously.
ECG monitoring during pacing must be performed with the ECG electrodes and patient
ECG cable. Pacing therapy electrodes cannot be used to monitor ECG rhythm and deliver
pacing current at the same time. Be sure to place the therapy electrodes in the proper
locations as described in the pacing procedure. Improper electrode placement may make
a difference in the capture threshold.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Press ON.
Instructor activity
Connect QUIK-
COMBO therapy
cable to the
simulator and
set rhythm to
bradycardia.
Task
Connect the patient
ECG cable, apply
ECG electrodes to
the ECG cable and
patient, and select
Lead I, II or III.
Identify the QUIK-
COMBO electrode
sites on the patient.
Prepare patient’s
skin for electrode
application.
User action, debrillator labels, text/voice
prompts and information
To receive the best monitoring signal, ensure there is
adequate space between the ECG electrodes and the
QUIK-COMBO electrodes.
For pacing, use either the anterior-lateral or anterior-
posterior position.
Task
Press the
PACER button.
Observe the
ECG rhythm.
User action, debrillator labels, text/voice
prompts and information
Conrm the PACER LED illuminates, indicating that
the power is on.
Note: If the REMOVE TEST PLUG message appears,
disconnect the test plug and connect therapy
electrodes to QUIK-COMBO therapy cable.
Conrm that a triangle sense marker appears near
the middle of each QRS complex. If the sense markers
do not appear or are displayed in the wrong location
(i.e.: T wave) select another lead.
Task
Press the
RATE button.
User action, debrillator labels, text/voice
prompts and information
• Turn SPEED DIAL (changes rate in increments
of 5 ppm) or press RATE button (changes rate in
increments of 10 ppm) to desired pacing rate.
• Pacing rate range from 40 to 170 ppm.
• Set rate to 80 ppm.

Instructor guide | 33
Task
Press the
CURRENT button.
User action, debrillator labels, text/voice
prompts and information
Turn SPEED DIAL (changes current in increments
of 5mA) or press CURRENT button (changes rate
in increments of 10mA) to increase current until
electrical capture occurs.
• For each delivered pacing stimulus, the PACER
indicator ashes off and a positive pace marker
displays on the ECG waveform.
• Pacing current range from 0 to 200mA.
• Many patients achieve capture at 50 to 100mA,
although individual thresholds vary. The simulator
achieves electrical capture at 65mA. Most
simulators achieves electrical capture between
65 and 75mA.
Task
Assess for
mechanical capture.
User action, debrillator labels, text/voice
prompts and information
Both electrical and mechanical capture must occur in
order for noninvasive pacing to benet the patient.
• Palpate patient’s pulse and obtain blood pressure
to assess for mechanical capture.
• Consider use of sedation/analgesia if necessary for
patient discomfort.
User action, debrillator labels, text/voice
prompts and information
Note: To interrupt pacing and view the patient’s
intrinsic rhythm, press and hold the PAUSE button.
This causes the pacer to pace at 25% of the set rate.
Release the PAUSE button to resume pacing at the set
rate. An ECG strip prints automatically for as long as
the button is held.
Task
To stop pacing.
User action, debrillator labels, text/voice
prompts and information
To stop pacing reduce current to zero or press PACER.
To debrillate and stop noninvasive pacing, press the
ENERGY SELECT button or charge the debrillator.
Pacing automatically stops.
Proceed with debrillation.
Instructor activity
Set simulator to VF
and demonstrate
debrillation
while pacing.
Discuss
Troubleshooting
• User observation.
• ECG leads off
during pacing.
User action, debrillator labels, text/voice
prompts and information
If the monitor detects ECG leads off during pacing,
pacing continues at a xed rate (nondemand pacing)
until the ECG lead is reattached. During xed-rate
pacing, the pacemaker delivers pulses at the set pace
rate regardless of any intrinsic beats that the patient
may have. The monitor continues to display the
pacing rate (ppm) and the current (mA). To reestablish
demand pacing, reattach the ECG lead.
While pacing, visually monitor the patient at all times,
do not rely on the ECG LEADS OFF warning to detect
changes in pacing function. Routinely assess the ECG
for proper sensing, pace pulse delivery, electrical and
mechanical capture
Task
QUIK-COMBO
electrodes off
during pacing.
User action, debrillator labels, text/voice
prompts and information
If the therapy electrodes detach during pacing,
the CONNECT ELECTRODES and PACING STOPPED
messages appear and an alarm sounds. The pacing
rate is maintained and the current resets to 0mA.
Reattaching the electrodes silences the alarm and
removes the CONNECT ELECTRODES message.
The current remains at 0mA until the current is
increased manually.
Instructor activity
Disconnect LL lead
from simulator

34 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
ECG monitoring
There are two methods for selecting or changing the ECG lead. Both methods are available
on your LIFEPAK 15 monitor/debrillator. The leads available depend on the ECG cable
(3-wire, 4-wire, 5-wire, or precordial leads) connected to the debrillator.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Press ON.
Instructor activity
Connect ECG
cable leads to
the simulator and
choose a rhythm
Task
Attach the ECG cable
to the monitor
Task
Identify the
appropriate electrode
sites on the patient
User action, debrillator labels, text/voice
prompts and information
Prepare the patient skin for electrode application:
• Remove excessive hair at electrode site.
Avoid placing electrodes over tendons
and major muscle masses.
• For oily skin, clean skin with an alcohol pad.
• Dry the site with a brisk rub.
Task
Apply ECG electrodes
User action, debrillator labels, text/voice
prompts and information
• Conrm package is sealed and use by date
has not passed.
• Attach an electrode to each lead wire.
• Grasp electrode tab and peel electrode from carrier.
• Inspect electrode gel for moisture content and to
conrm gel is intact.
• Apply the electrode at to skin. Smooth
tape outwardly. Avoid pressing the center
of the electrode.
• Secure the trunk cable clasp to the
patient’s clothing.
Task
Select the lead on the
monitor screen
User action, debrillator labels, text/voice
prompts and information
• Channel 1 op channel displays the primary
ECG waveform and is always visible when ECG
is displayed.
• Lead options available are dependent on which
ECG cable is used.
• Change lead by pressing the LEAD button and
select the desired lead with the SPEED DIAL or
by pressing the LEAD button.
• Adjust the ECG size by pressing the SIZE button
and select the size with the SPEED DIAL or by
pressing the SIZE button.
Task
Optional: Channel 2
and 3 waveforms
User action, debrillator labels, text/voice
prompts and information
• This can display an additional ECG waveform
or a continuation of the Channel 1 ECG.
• At the home screen, rotate the SPEED DIAL
to highlight Channel 2 or 3.
• Press the SPEED DIAL. An overlay appears with
the monitoring choice for the selected channel.
• Rotate and press the SPEED DIAL to select
monitoring choice.
Instructor guide | 35
Task
Adjusting the
Systole Tone Volume.
User action, debrillator labels, text/voice
prompts and information
Highlight and select heart rate (HR) in the monitoring
area of the screen.
• Rotate the SPEED DIAL to the desired volume.
• Press the HOME SCREEN to exit.
Discuss
Press PRINT
to obtain an
ECG printout.
User action, debrillator labels, text/voice
prompts and information
Prints continuously until you press the PRINT button
again to stop printing.
36 | Instructor guide for the LIFEPAK 15 monitor/debrillator/

Optional features

Instructor guide | 37
Acquiring a 12-lead ECG
The 12-lead electrocardiogram is used to identify, diagnose, and treat patients with cardiac
disorders and is useful in the early detection and prompt treatment of patients with acute
ST elevation myocardial infarction (STEMI).
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
To acquire a
12-lead ECG
Press ON.
Instructor activity
Connect the 12-lead
ECG cable leads to
the simulator and
choose a rhythm.
Task
Attach the ECG cable
to the monitor.
Task
Identify the
appropriate electrode
sites on the patient.
User action, debrillator labels, text/voice
prompts and information
Amplitude of the pulse bar indicates relative
signal strength.
• Remove excessive hair at electrode site.
Avoid placing electrodes over tendons and
major muscle masses.
• For oily skin, clean skin with an alcohol pad.
• Dry the site with a brisk rub.
Task
Apply ECG
electrodes.
User action, debrillator labels, text/voice
prompts and information
1. Conrm package is sealed and use by date
has not passed.
2. Attach an electrode to each lead wire.
3. Grasp electrode tab and peel electrode
from carrier.
4. Inspect electrode gel for moisture content
and to conrm gel is intact.
5. Apply the electrode at to skin. Smooth tape
outwardly. Avoid pressing the center of
the electrode.
6. Secure the trunk cable clasp to the
patient’s clothing.
Task
Press 12-lead.
Enter patients
age and sex.
User action, debrillator labels, text/voice
prompts and information
1. The 12-LEAD/AGE menu appears.
• Use the SPEED DIAL to select the age.
Note: Always enter the patient’s age if the patient
is 15 years old or younger. If you do not enter an
age, the default value of 50 years is used by the
interpretive analysis program and annotated on
the 12-lead ECG report.
2. The 12-LEAD/SEX menu appears.
• Use the SPEED DIAL to select the
patient’s sex.
Note: If you do not enter the sex, the default of male
is used by the interpretive analysis program and is
annotated on the 12-lead ECG report.
3. The monitor acquires, analyzes, and
automatically prints the 12-lead ECG.

38 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
SpO2, SpCO, and SpMet monitoring
SpO
2
, SpCO™, and SpMet™ are optional features for the LIFEPAK 15 monitor/debrillator.
When all three options (SpO
2
, SpCO, and SpMet) are installed, the pulse oximeter measures
functional oxygen saturation (SpO
2
), carboxyhemoglobin concentration (SpCO), and
methemoglobin concentration (SpMet) in the blood.
IMPORTANT! Masimo
®
Rainbow
®
sensors are necessary to monitor SpO
2
, SpCO, and
SpMet. These combination sensors as well as SpO
2
-only sensors are available for use
with the LIFEPAK 15 device. While Masimo SpO
2
-only sensors with red connector are
compatible with the LIFEPAK 15 device monitor, Masimo Rainbow sensors are not
compatible with other LIFEPAK debrillator/monitors.
Pulse Oximetry
EtCO
2
monitoring is used to detect trends in the level of expired CO
2
. It is used for
monitoring breathing efcacy and treatment effectiveness in acute cardiopulmonary care,
for example, to determine if adequate compressions are being performed during CPR or to
rapidly detect whether an endotrachael tube has been placed successfully.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Discuss
Turn the
debrillator ON.
User action, debrillator labels, text/voice
prompts and information
Press On.
Task
Connect the SpO
2
cable to the monitor.
User action, debrillator labels, text/voice
prompts and information
Attach the sensor to the SpO
2
cable and the patient..
• When the debrillator is turned on, the oximeter
turns on and performs a self-test that requires up
to 10 seconds.
• A sleep mode is activated within 10 seconds of
disconnecting the sensor.
• The oximeter will return to normal mode after
detecting a sensor or a patient signal.
Task
Observe the pulse
bar for uctuation.
User action, debrillator labels, text/voice
prompts and information
Amplitude of the pulse bar indicates relative signal
strength. the menu.
Task
Display waveform.
User action, debrillator labels, text/voice
prompts and information
• Select waveform channel 2 using the
SPEED DIAL.
• Select SpO
2
from the Waveform menu.
• The SpO
2
waveform automatically sizes itself
to provide optimum waveform viewing
Task
Adjust SpO
2
volume.
User action, debrillator labels, text/voice
prompts and information
4. Highlight and select SpO
2
on the home screen
with the SPEED DIAL:
5. Highlight and select SpO
2
VOLUME.
6. Rotate the SPEED DIAL to the desired volume.
7. Press the SPEED DIAL to set the volume.
Instructor guide | 39
Task
Adjust sensitivity.
User action, debrillator labels, text/voice
prompts and information
1. Highlight and select SpO
2
on the home
screen with the SPEED DIAL and then
select SENSITIVITY.
• Normal sensitivity is the default.
• High sensitivity allows monitoring in low
perfusion states, but is more susceptible
to artifact.
Task
Adjust averaging
time.
User action, debrillator labels, text/voice
prompts and information
2. Highlight and select SpO
2
on the home
screen with the SPEED DIAL and select
AVERAGING TIME.
3. Turn the SPEED DIAL to select and set
averaging time.
Options:
• 4 seconds (for patients with rapidly
changing values)
• 8 seconds (recommended for most patients)
• 12 and 16 seconds (when artifact is affecting
the performance of the pulse oximeter)
SpCO and SpMet
Monitoring SpCO and SpMet assists in identifying the often hidden conditions
of carboxyhemoglobinemia (carbon monoxide poisoning) and methemoglobinemia
(a condition that impedes delivery of oxygen to the tissues). Low levels of both SpCO
and SpMet are normally found in the blood; however, early detection of signicantly high
levels can lead to proper diagnosis and treatment and can help improve patient outcome.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Turn the
debrillator ON.
User action, debrillator labels, text/voice
prompts and information
Press ON.
Task
Connect the
Rainbow SpO
2
cable to the monitor.
User action, debrillator labels, text/voice
prompts and information
Attach the Rainbow sensor to the SpO
2
cable
and the patient.
Task
Verify that an
SpCO/SpMet sensor
is in use. Only
Rainbow sensors
are capable of
reading SpCO/SpMet.
User action, debrillator labels, text/voice
prompts and information
• When the debrillator is turned on, the oximeter
turns on and performs a self-test that requires up
to 20 seconds.
• A sleep mode is activated within 10 seconds of
disconnecting the sensor.
• The oximeter will return to normal mode after
detecting a sensor or a patient signal.
Task
Press PRINT to
obtain SpCO
or SpMet value.
User action, debrillator labels, text/voice
prompts and information
Press PRINT
• If dashes (---) appear on printout instead of values
for SpCO or SpMet, allow a few more seconds for
measurement to be obtained.
40 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Task
Display SpCO
or SpMet value.
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to select the SpO
2
area.
2. Select PARAMETER from menu.
3. Select SPCO or SPMET. Selected value will
display for 10 seconds then revert to SpO
2
.
Task
SpCO/SpMet
Advisory
If the SpCO or SpMet reading is above normal limits,
indicating a dangerous amount of carboxyhemoglobin
or methemoglobin, an Advisory occurs.
During an Advisory:
• The elevated SpCO or SpMet value is displayed
instead of SpO
2
.
• The elevated value ashes and the alarm
tone sounds.
• One of the following Advisory messages
appears in the message area:
• Advisory: SpCO > 10%
• Advisory: SpMet > 3%
Keys to successful SpCO
and SpMet monitoring
Because of the increased sensitivity of SpCO and SpMet monitoring, extra care is needed
to ensure the sensor will function appropriately. Careful attention to sensor placement
and protection from ambient light is particularly important, as well as other identied
solutions listed below.
Challenges Solutions
Low arterial perfusion Choose a site that is well perfused (i.e., the warmest extremity).
Conrm BP cuff is on opposite extremity.
Motion Place sensor on ring nger of non-dominant hand and restrict
patient movement.
Consider adhesive sensor if available.
Poor sensor placement Orient the sensor so the cable is on the back of the patient’s hand. The
tip of the nger should touch the raised digit stop inside the sensor.
Reposition sensor as needed.
Finger nail polish Always remove polish.
Strobe or ashing light Cover sensor with opaque material to protect from light.
Unexpected readings In addition to above troubleshooting methods, take readings on
3 separate digits and average number.
Ambient light Cover sensor with opaque material to protect from light.
Debrillation Wait for approx 20 seconds to recalibrate.
Slender digits Use on largest digit such as the index nger.
No SpCO or SpMet
value (---) is displayed
In addition to the above listed troubleshooting, if values do not
display within 30 seconds, disconnect and reconnect sensor.
Failure to apply the sensor properly may cause incorrect measurements.
SpCO accuracy during no motion for 1 to 40% is ±3% (1 SD) for adults and pediatrics.
SpMet accuracy during no motion for 0 to15% ±2%.

Instructor guide | 41
Monitoring noninvasive blood pressure
The LIFEPAK 15 device noninvasive blood pressure (NIBP) monitor measures blood
pressure (BP) using the oscillometric measurement technique to determine systolic,
diastolic, and mean arterial pressures and pulse rate. The measurement can be initiated
manually or set to recur automatically at predetermined intervals.
Noninvasive blood pressure monitoring is intended for detection of hypertension or
hypotension and monitoring BP trends in patient conditions such as, but not limited to,
shock, acute dysrhythmia or major uid imbalance.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
To obtain a manual
single measurement:
Press ON.
User action, debrillator labels, text/voice
prompts and information
1. Apply appropriately sized cuff and properly
align cuff artery markings to extremity.
2. Connect tubing to cuff and NIBP port
on the monitor.
3. Position extremity in relaxed and supported
position at heart level.
4. Inform patient that the cuff will inate
and squeeze arm.
5. Press NIBP to start measurement.
To cancel a measurement in progress,
press NIBP again.
Task
To obtain a
time controlled
measurement:
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to outline NIBP area.
2. Press the SPEED DIAL.
3. Select INTERVAL.
4. Rotate the SPEED DIAL to select the desired
time interval.
5. Press the SPEED DIAL to set the time interval.
6. Press NIBP to start measurement.
Task
To change
in initial
measurement
pressure:
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to outline NIBP area.
2. Press the SPEED DIAL.
3. Select INITIAL PRESSURE.
4. Rotate the SPEED DIAL to the desired pressure.
5. Press the SPEED DIAL to set the initial interval.
6. Press NIBP to start measurement.

42 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Monitoring EtCO2
The end-tidal CO
2
(EtCO
2
) monitor is a capnometric device that uses non-dispersive
infrared spectroscopy to continuously measure the amount of CO
2
during each breath and
report the amount present at the end of exhalation (EtCO
2
). The sample is obtained by the
side stream method and can be used with intubated or nonintubated patients. Respiration
rate is also measured and displayed in breaths per minute.
EtCO
2
monitoring is used to detect trends in the level of expired CO
2
. It is used for
monitoring breathing efcacy and treatment effectiveness in acute cardiopulmonary
care, for example, to determine if adequate compressions are being performed during
CPR or to rapidly detect whether an endotrachael tube has been placed successfully.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
To monitor EtCO
2
:
Press ON.
User action, debrillator labels, text/voice
prompts and information
1. S
elect the appropriate EtCO
2
accessory
for the patient.
2. Open the CO
2
port door and insert the
FilterLine connector; turn connector
clockwise until hand tight.
3. Verify that the CO
2
area is displayed. The EtCO
2
monitor performs the autozero routine as part of
the initialization self-test.
4. Display CO
2
waveform in Channel 2 or 3.
5. Connect the CO
2
FilterLine set to the patient.
6. Confirm that the EtCO
2
value and waveform are
displayed. The monitor automatically selects the
scale for the best visualization of the waveform.
Task
To display the
EtCO
2
waveform.
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to outline display
channel 2 or 3.
2. Press the SPEED DIAL.
3. Select WAVEFORM.
4. Rotate the SPEED DIAL to select CO
2
.
5. Press the SPEED DIAL.
6. Press the HOME SCREEN button to
clear the menu.
Task
To change the
CO
2
scale:
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to outline CO
2
area.
2. Press the SPEED DIAL.
3. Select SCALE.
4. Rotate the SPEED DIAL to select the
desired scale.
• Autoscale (default)
• 0–20 mmHg (0–4 Vol% or kPa)
• 0–50 mmHg (0–7 Vol% or kPa)
• 0–100 mmHg (0–14 Vol% or kPa)
5. Press the SPEED DIAL to set the scale.
6. Press the HOME SCREEN button to clear
the menu.

Instructor guide | 43
Monitoring Invasive Pressure
The LIFEPAK 15 device invasive pressure (IP) monitor is intended for measuring arterial,
venous, intracranial and other physiological pressures using an invasive catheter system
with a compatible transducer.
Invasive pressure monitoring is indicated for use in patients who require continuous
monitoring of physiological pressures in order to rapidly assess changes in the patient’s
condition or response to therapy. It may also be used to aid in medical diagnosis.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
To monitor
Invasive Pressures
Press ON.
User action, debrillator labels, text/voice
prompts and information
1. Prepare the transducer system according to
the operating instructions provided with the
transducer and your local protocol.
2. Connect the IP cable to the transducer and to
the P1 port on the monitor.
3. Use the default label P1 or select ART, PA, CVP,
ICP, or LAP. To change the label, select the P1
area. From the menu, select P1. Select a label
from the list.
4. Use the SPEED DIAL to outline and select
CHANNEL 2 on the Home Screen. From the
Channel 2 menu, select WAVEFORM and then
select the label that is desired for the waveform.
5. Open the transducer’s stopcock to air to zero
the transducer and remove stopcock cap. Select
the P1 area. Select ZERO from the menu. The
message P1 ZEROED appears when zeroing is
complete and the pressure values are displayed
as zeros.
6. Close the stopcock to air. The patient’s pressure
waveform should be displayed. A scale is
automatically selected to display the pressure.
Conrm that pressure amplitude correlates with
the digital readout.
Note: If you place a cap on an open port before you
close the port to air, an error message may appear.
You will be required to zero the transducer again.
Task
To display the
EtCO
2
waveform.
User action, debrillator labels, text/voice
prompts and information
The IP monitor can display pressures from -30 to 300
mmHg. After zeroing the transducer pressure, the
monitor automatically selects one of the following
scales based on the patient’s measured pressure:
• -30 to 30 mmHg
• 0 to 60mmHg
• 0 to 120 mmHg
• 0 to 150 mmHg
• 0 to 180 mmHg
• 0 to 300 mmHg
You can also manually select one of these scales
or autoscale to readjust the waveform within
the channel.
Task
To change the
pressure scale
User action, debrillator labels, text/voice
prompts and information
1. Use the SPEED DIAL to outline and select the
P1 area. The P1 menu appears.
2. From the menu, select SCALE and then choose
a scale from the list.

44 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Monitoring continuous temperature
The LIFEPAK 15 device temperature monitor is intended for use in patients who require
continuous monitoring of body temperature.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
To Monitor
Continuous
Temperature:
Press ON.
User action, debrillator labels, text/voice
prompts and information
1. Connect adapter cable to TEMP port on monitor.
2. Connect temperature probe to adapter cable.
3. Place temperature probe on patient according
to probe Instructions for Use.
4. Conrm temperature reading appears
and is stable.
Vital signs and ST segment trends
The trends feature of the LIFEPAK 15 monitor/debrillator provides the ability
to graphically display and document the patient’s vital signs (VS) and ST segment
measurements for up to eight hours. VS trending is intended for use with any patient who
requires continuous monitoring of vital signs over an extended period of time to identify
changes in patient condition and to document patient response to therapy. ST trending
is intended for use with patients suspected of having acute ischemic events, such as
unstable angina, and for patients during treatment of an acute ischemic event. ST segment
measurement is initiated using a 12-lead ECG and is derived using the University of
Glasgow 12-Lead ECG Analysis Program.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
How VS trends work
User action, debrillator labels, text/voice
prompts and information
• Each active vital sign can be displayed
graphically for time ranges of 30 minutes,
and 1, 2, 4, and 8 hours.
• The vital signs are HR, SpO
2
, SpCO, SpMet, CO
2
,
and RR; and systolic, diastolic and mean pressures.
• Data is sampled every 30 seconds. If valid data
is not available, a blank space is substituted
on the graph.
• NIBP values are plotted only when an NIBP
measurement is obtained.
• VS measurements are not averaged or ltered.
• No messages or alarms occur based on changes
in VS measurements.

Instructor guide | 45
Task
How ST trends work
User action, debrillator labels, text/voice
prompts and information
• ST measurements can be displayed graphically for
time ranges of 30 minutes, and 1, 2, 4, and 8 hours.
• ST trending is initiated by obtaining the patient’s
rst 12-lead ECG.
• The ST J-point (STJ) measurement is plotted on
the ST trend graph.
• When all leads of the 12-lead ECG cable are
attached to the patient, STJ measurements are
obtained automatically every 30 seconds.
• If a lead is off, or the ECG data is too noisy, ST
measurements are not obtained and the graph
shows a blank for that time period.
• If an STJ measurement in any lead deviates from
the initial measurement by 1 mm (0.1 mV) or more
and the deviation persists for 2.5 minutes, the
monitor automatically prints another 12-lead ECG.
Task
To display
trend graphs:
User action, debrillator labels, text/voice
prompts and information
1. Rotate the SPEED DIAL to outline Channel 2 or
3, and then press the SPEED DIAL to select the
channel. The Channel menu appears.
2. Select WAVEFORM, and then select TREND.
3. Select SOURCE, and then select the desired
VS or ST.
• The default setting for SCALE and
RANGE is AUTO.
• When AUTO is used, the monitor
automatically updates the scale so that
all values are displayed and all data from
Power On to the present time is visible.
• If you change scale or range, some data
may not be visible because it is off scale
or out of range.
4. Press HOME SCREEN. The graph for the
selected VS or ST appears in the channel.
Note: To initiate ST trends, you must obtain a
12-lead ECG. The initial ECG provides the baseline
ST measurement and initiates the ST trends feature.
Task
To print
trend graphs:
User action, debrillator labels, text/voice
prompts and information
1. Press OPTIONS. The Options menu appears.
2. Rotate and then press the SPEED DIAL
to select PRINT.
3. Select REPORT, and then select
TREND SUMMARY.
4. Select PRINT. The Trend Summary Report
prints graphs of all actively monitored
VS and ST trends.
46 | Instructor guide for the LIFEPAK 15 monitor/debrillator/

Data management
and other features

Instructor guide | 47
Data management
When you turn on the LIFEPAK 15 monitor/debrillator, you create a new Patient Record
stamped with the current date and time. All events and associated waveforms are digitally
stored in the Patient Record as patient reports. When you turn the device off, the current
Patient Record data is saved in the patient archives.
The Patient Record can be printed for storage in the patient’s paper le. It can also be
uploaded and reviewed on a computer with CODE-STAT™ Data Review Software with
Advanced CPR Analytics. This feature allows the user to collect, manage and analyze
postevent CPR performance and can help your hospital manage quality assurance and
improve responder performance.
Hospitals and EMS services have different approaches to managing patient data.
The training for your staff will depend on your particular approach to collecting
and storing patient data.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
EVENT
User action, debrillator labels, text/voice
prompts and information
• Pressing the EVENT button displays a menu
showing drug names or activities that may have
been given or done during the debrillator use.
• Use the SPEED DIAL to scroll through and
select the menu choices. The selected event
and time stamp appear in the message area
on the screen and are printed in the CODE
SUMMARY Event Log.
Task
CODE SUMMARY
Critical Event Record
User action, debrillator labels, text/voice
prompts and information
A CODE SUMMARY report is automatically stored as
part of the patient record for each patient. The report
consists of:
• Preamble Contains patient demographic and
device information. The event identication
composed of the date and time the debrillator is
turned on, is automatically entered in the ID eld.
• Event/Vital Signs Log Contains events and vital
signs in chronological order. Events are device or
operator actions that are related to the use of the
debrillator. Vital signs (HR, EtCO
2
, SpO
2
, SpCO,
SpMet, NIBP, Temperature, IP as applicable) are
entered into the log automatically every 5 minutes.
• Waveform Events Therapy and other selected
events also capture waveform data. Refer to the
Operating Instructions for more detail.
Instructor activity
Press CODE
SUMMARY to print
a code summary for
the current patient.
Task
Managing Archived
Patient Records
Press OPTIONS.
Select ARCHIVES.
User action, debrillator labels, text/voice
prompts and information
Data Storage: When the LIFEPAK 15 device is turned
on, a new patient record is created. The report is
automatically stamped with the patient ID and
is saved in ARCHIVES when the debrillator
is turned off.
• When Archives is entered, patient monitoring ends
and the current patient record is saved and closed.
• Print Allows printing of CODE SUMMARY for
selected patient.
• Edit Allows editing of selected elds in the patient
record such as name, ID, sex, etc.
• Delete Allows deletion of selected patient records.
To exit Archives, turn off the debrillator.

48 | Instructor guide for the LIFEPAK 15 monitor/debrillator/
Discuss
Memory Capacity
User action, debrillator labels, text/voice
prompts and information
The LIFEPAK 15 device retains data for two or more
patients when you switch the power off. The number
of patient reports stored depends on various factors,
including the number of displayed waveforms,
the duration of each use, and the type of therapy.
Typically, memory capacity includes up to 100 single
waveform reports. When memory capacity is reached,
the debrillator deletes an entire Patient Record using
a “rst in, rst out” priority. Deleted records cannot
be retrieved.
Discuss
Uploading data
to CODE-STAT
with Advanced
CPR Analytics
User action, debrillator labels, text/voice
prompts and information
Patient reports and data can be easily downloaded
from your LIFEPAK 15 device to a computer that has
either the CODE-STAT Data Review Software with
Advanced CPR Analytics or DT EXPRESS™ Data
Transfer Software loaded on it. A download wizard
makes this process simple and quick once the software
has been loaded on the computer. This feature allows
the user to collect, manage, and analyze post-event
CPR performance.
Task
Printing a record
User action, debrillator labels, text/voice
prompts and information
1. Press PRINT to turn on printer.
• Printout 8 second delay.
• Printout will include channels displayed
on screen.
2. Press PRINT to turn off printer.

Instructor guide | 49
Other functions
Setup options allow you to dene operating features for the LIFEPAK 15 monitor/
debrillator such as device identication numbers and default settings.
References to buttons are indicated in BOLD and display messages are indicated in
ITALICS. For complete information, review the Operating Instructions for the device.
Task
Setting alarms
User action, debrillator labels, text/voice
prompts and information
1. Press ALARMS.
2. Select QUICK SET to activate the alarms for all
active parameters (HR and SpO
2
if applicable).
3. Select LIMITS to set or change the alarm limits
to WIDE or NARROW.
• Limits are set based on the patient’s current
HR and parameter values.
4. Select SUSPEND to turn off the audible alarm
tone for up to 15 minutes.
5. Select VF/VT ALARM to turn on continuous
monitoring for ventricular brillation and
ventricular tachycardia in manual mode.
A symbol (magnifying glass) appears above the
primary ECG when the alarm is on.
Task
Managing alarms
User action, debrillator labels, text/voice
prompts and information
The alarm bell symbol indicates when alarms
are ON or OFF.
• When alarms are OFF a red X appears
over the bell.
• When alarms are ON and an alarm limit is
exceeded, a tone sounds, the violated parameter
ashes, and an alarm message appears.
To manage an alarm:
1. Press ALARMS. This silences the alarm tone
for 2 minutes.
• Assess the cause of the alarm.
• Assess the appropriateness of the limits
setting (WIDE or NARROW).
2. If the patient is unstable, consider suspending
the alarm tone for up to 15 minutes. Do not
reselect QUICK SET.
3. Once the patient is stable reselect QUICK SET
if necessary.
Task
Options
User action, debrillator labels, text/voice
prompts and information
• Pressing OPTIONS displays a menu showing
the following items.
• Use the SPEED DIAL to scroll through and
select the menu choices.
• PATIENT Allows entering of patient demographic
information into the patient record.
• PACING Selects demand or nondemand pacing
and internal pacer detection on or off.
• PRINT Allows printing of
CODE SUMMARY reports.
• ARCHIVES Accesses archived patient records.
• DATE/TIME Sets the date and time. For changes
to take effect, cycle power.
• ALARM VOLUME Adjusts volume for alarms,
tones, and voice prompts.
• USER TEST Initiates device testing.
50 | Instructor guide for the LIFEPAK 15 monitor/debrillator/

Instructor guide | 51
LIFEPAK
®
15 Monitor/Debrillator
BRIEF SUMMARY OF INDICATIONS AND IMPORTANT SAFETY INFORMATION
LIFEPAK 15 is a complete acute cardiac care response system designed for basic life support (BLS) and advanced life
support (ALS) patient management protocols. INTENDED USE: LIFEPAK 15 intended for use by trained medical
personnel out-of-doors, in indoor emergency care settings, and is designed to be used for ground transportation.
Monitoring and therapy functions may only be used on one patient at a time. Manual mode monitoring and therapy
functions are intended for use on adult and pediatric patients. Automated external debrillation (AED) mode intended
for use on patients ≥ 8 years of age.
INDICATIONS FOR USE – MANUAL DEFIBRILLATION: Indicated for termination of certain potentially fatal
arrhythmias, such as ventricular brillation and symptomatic ventricular tachycardia. Delivery of energy in
synchronized mode is a method for treating atrial brillation, atrial utter, paroxysmal supraventricular tachycardia
and, in relatively stable patients, ventricular tachycardia. CONTRAINDICATIONS - MANUAL DEFIBRILLATION:
Contraindicated in treatment of PEA and asystole. AED MODE: To be used only on patients in cardiopulmonary arrest.
Patient must be unconscious, pulseless, and not breathing normally before using debrillator to analyze patient’s ECG
rhythm. In AED mode, the LIFEPAK 15 is intended for use on pediatric patients ≥ 8 years of age.
CONTRAINDICATIONS - AED MODE: None known.
INDICATIONS FOR USE – MONITORING. AQUIRING 12-LEAD ECG: 12-lead electrocardiogram used to identify,
diagnose, and treat patients with cardiac disorders and is useful in early detection and prompt treatment of patients
with STEMI. MONITORING SPO
2
, SPCO, AND SPMET: Pulse oximetry indicated for use in any patient who is at
risk of developing hypoxemia, carboxyhemoglobinemia, or methemoglobinemia. SpO
2
monitoring may be used during
no motion and motion conditions, and in patients who are well or poorly perfused. SpCO and SpMet accuracies have
not been validated under motion or low perfusion conditions. MONITORING NONINVASIVE BLOOD PRESSURE:
Intended for detection of hypertension or hypotension and monitoring blood pressure trends in patient conditions.
NIBP monitoring not indicated for neonatal patients <1-month-old. MONITORING EtCO
2
: Used to detect trends in
level of expired CO
2
, used for monitoring breathing efcacy and treatment effectiveness in acute cardiopulmonary
care. MONITORING INVASIVE PRESSURE: Indicated for use in patients who require continuous monitoring of
physiological pressures to rapidly assess changes in patient’s condition or response to therapy. May also be used to aid
diagnosis. MONITORING CONTINUOUS TEMPERATURE: Indicated for use in patients who require continuous
monitoring of body temperature.
MONITORING CONTRAINDICATIONS: None known.
Operating Instructions provide important information to help you operate LIFEPAK 15. Become familiar
with all terms and warnings. GENERAL DANGER: Explosion hazard. GENERAL/THERAPY/MANUAL
DEFIBRILLATION WARNINGS and CAUTION: Shock or re hazards • Possible patient skin burns and ineffective
energy delivery • Possible device failure, damage, inability to deliver therapy, shutdown, loss of power during patient
care, improper device performance • Possible electrical interference with device performance or with other equipment
• Safety risk • Failure to detect change in ECG rhythm • Possible failure to detect out of range condition • Possible
interference with implanted electrical device • Possible paddle damage • Possible incorrect energy delivery. CPR
METRONOME WARNING: CPR delivered when not needed. SYNCHRONIZED CARDIOVERSION WARNING:
Possible lethal arrhythmia. NONINVASIVE PACING WARNING: Possible inability to pace, interruption of therapy,
ineffective pacing, and patient skin burns. PEDIATRIC ECG MONITORING AND MANUAL MODE THERAPY:
Possible patient skin burns. AED WARNINGS: Possible misinterpretation of data or ECG misinterpretation •
Pediatric patient safety risk. ECG MONITORING WARNING: Possible misinterpretation of ECG data. 12-LEAD ECG
WARNINGS: Possible inability to obtain diagnostic quality 12-lead ECG or inaccurate diagnosis • Possible incorrect
treatment with reperfusion therapy. SPO
2
, SPCO, AND SPMET WARNINGS AND CAUTION: Shock or burn hazard
• Inaccurate pulse oximeter readings • Possible skin injury • Possible strangulation • Inaccurate SPO
2
, SPCO and/
or SPMET readings • Possible equipment damage. NIBP MONITORING WARNINGS AND CAUTION: Possible loss
of IV access and inaccurate infusion rate, circulation impairment or inaccurate blood pressure or oxygen saturation
readings • Possible patient harm • Equipment damage. EtCO
2
MONITORING WARNINGS AND CAUTION: Fire
hazard • Possible inaccurate patient assessment or inaccurate CO
2
readings • Possible strangulation • Infection
hazard • Possible equipment damage. IP MONITORING WARNINGS: Possible inaccurate pressure readings, air
embolism, blood loss or loss of sterility • Possible patient injury or equipment damage • Possible lethal arrhythmia
• Increased intracranial pressure. TEMPERATURE MONITORING WARNINGS: Possible inaccurate temperature
readings • Infection hazard • Possible strangulation. VITAL SIGN/ST SEGMENT TRENDS WARNING: Inaccurate
interpretation of patient status.
U.S. Federal law restricts this device to sale by or on the order of a physician.
Please consult Operating Instructions at www.strykeremergencycare.com or call 800.442.1142 for complete list of
indications, contraindications, warnings, cautions, potential adverse events, safety and effectiveness data, instructions
for use and other important information.
For further information, please contact your
Stryker representative or visit our website at
strykeremergencycare.com
Emergency Care
This document is intended solely for the use of healthcare professionals. A healthcare
professional must always rely on his or her own professional clinical judgment when
deciding whether to use a particular product when treating a particular patient. Stryker
does not dispense medical advice and recommends that healthcare professionals be
trained in the use of any particular product before using it.
The information presented is intended to demonstrate Stryker’s product offerings.
A healthcare professional must always refer to operating instructions for complete
directions for use indications, contraindications, warnings, cautions, and potential
adverse events, before using any of Stryker’s products. Products may not be available
in all markets because product availability is subject to the regulatory and/or medical
practices in individual markets. Please contact your representative if you have questions
about the availability of Stryker’s products in your area. Specications subject to change
without notice.
Stryker or its afliated entities own, use, or have applied for the following trademarks or
service marks: CodeManagement Module, CODE-STAT, CODE SUMMARY, DT EXPRESS,
EDGE System, LIFEPAK, QUIK-COMBO, REDI-CHARGE, REDI-PAK, Stryker, SunVue. All
other trademarks are trademarks of their respective owners or holders.
The absence of a product, feature, or service name, or logo from this list does not
constitute a waiver of Stryker’s trademark or other intellectual property rights
concerning that name or logo.
07/2020
GDR 3309799_D
Copyright © 2020 Stryker
References
1. Chan PS, Krumholz HM, Nichol G, Nallamothu BK. “Delayed time to debrillation after
in-hospital cardiac arrest.” NEJM. 2008;358:9–17.
2. Stiell IG, Walker RG, Nesbitt LP, et al. “The BIPHASIC Trial: A randomized comparison of xed
lower versus escalating higher energy levels for debrillation in out-of-hospital cardiac arrest.”
Circulation. 2007;115:1511–1517.
3. Koster RW, Walker RG, Chapman F. “Recurrent ventricular brillation during advanced life
support care of patients with prehospital cardiac arrest.” Resuscitation. 2008;78:252–257.
4. Walsh SJ, McClelland AJ, Owens CG, et al. “Efcacy of distinct energy delivery protocols
comparing two biphasic debrillators for cardiac arrest.” Am J Cardiology. 2004;94:378–380.
Manufactured by:
Physio-Control, Inc.
11811 Willows Road NE
Redmond, WA, 98052 U.S.A.
Toll free 800 442 1142
strykeremergencycare.com
Stryker European
Operations B.V.
Herikerbergweg 110
1101 CM Amsterdam
Netherlands
Tel +31 (0)43 3620008
Fax +31 (0)43 3632001
Distributed by:
Stryker Canada
2 Medicorum Place
Waterdown, Ontario
L8B 1W2
Canada
Toll free 800 668 8323
Stryker UK Ltd
Stryker House
Hambridge Road
Newbury, Berkshire
RG14 5AW
United Kingdom
